|
11/22/2023 0 Comments Can entropy decrease Explanation: Entropy (S) by the modern definition is the amount of energy dispersal in a system. Entropy doesn't simply disappear, so the only way to decrease the entropy of the universe would be to move it outside of the universe, but that idea is way beyond my undergraduate level understanding of. The system entropy (S) by the modern definition is the amount of energy dispersal in a system. The entropy of a system can decrease, but only by a transfer of an equivalent amount of entropy, or more, from that system to another system. Even if it is true that the processes of life on earth result in an entropy decrease of the earth, the second law of thermodynamics will not be violated unless that decrease is larger than the entropy increase of the two heat reservoirs. Apart from a factor like a change in the number of molecules involved, internal entropy changes are often fairly subtle. 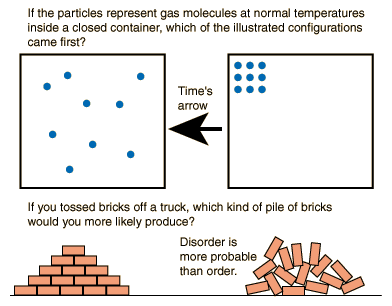
Entropy generally decreases when a reaction produces fewer molecules than it started with. For processes in which the temperature is not constant such as heating or cooling of a substance, the equation must be integrated over the required temperature range, as discussed below. When does entropy increase and when does it decrease Chemistry Thermochemistry Entropy 1 Answer Dr. Entropy generally increases when a reaction produces more molecules than it started with. Now I understand why the entropy can't decrease, but I fail to understand why the entropy tends to increase as the system reach the thermodynamic. This is the basic way of evaluating \(ΔS\) for constant-temperature processes such as phase changes, or the isothermal expansion of a gas. The second law of thermodynamics states that the entropy of an isolated system never decreases, because isolated systems always evolve toward thermodynamic equilibrium, a state with maximum entropy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
